Iron Giant?
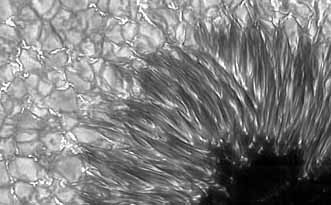
Here is an amazing image (using adaptive optics) of the huge sunspot that crossed old Sol at the end of September. Kind of makes you wonder what the heck our Sun is made of.
Which reminds Top Men at Wohba that there is a growing movement that is taking issue with the traditional giant-ball-of-gas model of the sun first posited by Galileo. The leading advocates claim that much of the unexplained solar phenomenon begins to make perfect sense with a solid sun model. Some big-thinkers are even advancing the idea that the sun is substantially iron - with a swirling liquid-like plasma ocean of neon covering the surface. Some other big-thinkers think those first big-thinkers are smokin' something.
This web site presents the case. Here is another for reference.
Top Men are always amazed at how much we know, and how much we don't.




2 Comments:
The Sun acts as a magnetic plasma diffuser, selectively moving lighter atoms to its surface.
http://tinyurl.com/3ydcql
The most abundant elements inside the Sun are the same elements that are most abundant in ordinary meteorites and in planets close to the Sun: Iron, oxygen, silicon, nickel, and sulfur.
http://tinyurl.com/2rwvvh
Successively lighter elements are enriched at the Sun's surface by factors of 10; 100; 1,000; 10,000; 10,000; 100,000; and even 1,000,000 !
The lightest element is hydrogen. The next lightest one is helium.
The solar surface is 91% hydrogen and 9% helium. The other 81 elements make up only 0.2% of the elements at the Sun's surface.
Solar flares partially disrupt mass separation. On 10 Sept 2000, NASA reported that successively heavier elements were enriched in a solar flare by factors of ~10, ~100, and ~1000.
http://tinyurl.com/2tjjtx
On 20 July 2004, NASA found that the heaviest elements in solar flares were enriched by as much as ~10,000.
http://tinyurl.com/2q4wvb
Here is other evidence that the model of a hydrogen-filled is obsolete.
http://arxiv.org/pdf/astro-ph/0410569
With kind regards,
Oliver
www.omatumr.com
In the July 5, 2007 issue of Nature, astronomer John Cowan [1] and the editor [2] discuss another "surprise" in astronomy: The abundance pattern of heavy elements formed by neutron-capture in the oldest stars in our Galaxy -- "the Methuselahs of the star world" -- mirror those in "infants such as the our Sun".
These observations surprised those who assumed that:
a.) A "Big Bang" made the universe and all its hydrogen,
b.) The hydrogen-rich surface layer of a star represents its internal composition, and
c.) Heavy elements like xenon, barium, and europium were made by neutron-capture reactions in stars millions or billions of years after the Big Bang.
However, these assumptions are falsified and mass separation of elements in the Sun [3] is confirmed by comparing the abundance pattern of heavy elements formed by neutron-capture with that predicted from the well-known cross sections for neutron-capture reactions [4, 5].
1. "Astronomy: A constant surprise," Nature 448 (5 July 2007) p. 2.
http://www.nature.com/nature/journal/v448/n7149/full/448029a.html
2. "Stars in their element", Editor's summary, Nature 448 (5 July 2007).
http://www.nature.com/nature/journal/v448/n7149/edsumm/e070705-05.html
3. "Solar abundance of the elements", Meteoritics 18 (1983) pp. 209-222.
http://www.omatumr.com/archive/SolarAbundances.pdf
4. "Solar abundance of elements from neutron-capture cross sections," 36th Lunar and Planetary Science Conference (2005), paper 1033
http://www.omatumr.com/abstracts2005/LunarAbstract.pdf
5. "Nuclear Systematics: Part IV. Neutron-capture cross sections and solar abundance", J. Radioanalytical Nuclear Chemistry 266 (2005) pp. 159–163.
http://www.omatumr.com/abstracts2005/Fk01.pdf
CONCLUSION: Isotope ratios from the 1969 Apollo Mission to the Moon revealed evidence of severe mass separation in the Sun, over the mass range of 3 to 136 times the mass of the Hydrogen atom.
Dramatic similarities in the relative abundance patterns of heavy elements made by neutron-capture at the surfaces of stars of various ages, over the mass range of 25 to 207 times the mass of the Hydrogen atom, confirm that mass separation masks the iron-rich interiors of most stars with a veneer of the lightest element, Hydrogen.
With kind regards,
Oliver K. Manuel
www.omatumr.com
Post a Comment
<< Home